

Asterionella formosa: a delicate microalga perfect for cosmetics
October 6, 2020
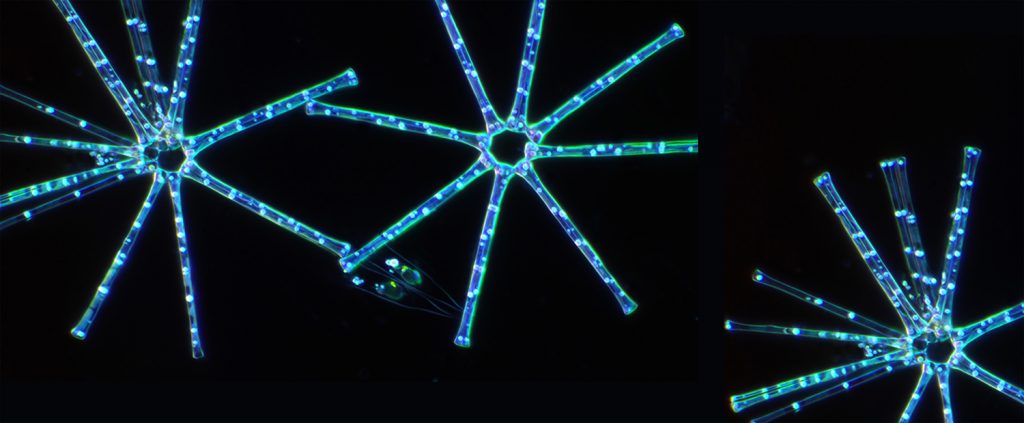
Let’s delve deeper into Asterionella formosa, a microalga that stands out from the crowd with exceptional advantages for cosmetic products.
Asterionella formosa, a diatom (brown) microalga, has a lot to offer us and our wellbeing. Unlike other Asterionella, A. formosa evolves in temperate climates, preferably in fresh water at 10-15°. But it is very good at adapting – one of its main advantages. Measuring no more than 60 micrometers (0.06 mm), this “colonial” microalga can reproduce in much lower temperatures, as well as in environments where nutrients are scarce.
Another big advantage? Asterionella formosa is a biomarker that identifies negative human impacts on the environment like the eutrophication of lakes (Asterionella proliferates) and their acidification (Asterionella populations decrease). It is therefore invaluable for Paleoecology and Paleolimnology[1] experts looking to reconstruct the sedimentary past of inland waters.
The microalga with multiple benefits for cosmetics
Cosmetics industry professionals are keeping a close eye on this microalga. The external structure is rich in biogenic silica, a trace element essential for protecting and reinforcing the skin, hair and nails. It also produces:
- Fucoxanthine, a molecule with antioxidant, anti-inflammatory and fat-burning properties
- Long-chain polyunsaturated omega 3 fatty acids essential to human health
- A, B and C asterionelines with anti-inflammatory properties
- Phycosterols (β-sitosterol) that act as anti-inflammatory, antioxidant, cholesterol-regulating agents
Fragile yet strong
It is no coincidence that Micophyt chose the microalga’s star-shaped structure for its logo. This reference alludes to the top-ranking French startup’s technological capacity to develop even the most delicate microalgae on a large scale. As is the case for Asterionella formosa. “Very few are able to cultivate it in an artificial environment that enables mass production. Its fragility and nutritional needs require adapted technology,” explains Rémi Pradelles, Microphyt’s Research & Development Director. In 2015, the company included Asterionella formosa as one of the 40 most promising microalgae in the SMILE[2] project, launched in collaboration with CEA. The aim? Observe how these microalgae grow in a closed environment.
Innovative technology to get the best from microalgae
In Microphyt’s laboratory, Asterionella formosa has unveiled many of its secrets, in particular its metabolic growth levers. The next step? Producing it in a controlled, standardized environment in industrial conditions. And, this is where Camargue® technology – a unique production process designed and patented by Microphyt – comes in. The 5000-liter tubular photo-bioreactors recreate smooth wave patterns while also preserving the cellular integrity of fragile microalgae including Asterionella formosa. This is excellent news for anyone looking to co-develop differentiating cosmetic products based on a microalga that actively promotes wellbeing.

[1] Paleolimnology refers to studying the history of inland waters (including lakes, steams and wetlands) by analyzing sedimentary deposits.
[2] The SMILE (Slimming and Memory booster mIcroaLgae Extract) program aims to develop, produce and commercialize active natural ingredients extracted from specific microalgae that contribute to weight loss and maintaining cognitive functions. This program is supported by the European Union.




